Ozone is used in paper mills for pulp bleaching and decolorization, and it has the ability to decolorize all dyeing wastewater. Ozone can break down the chromogenic and assisting groups of these dyes, thereby achieving decolorization effects, but the effect of ozone on various organic dyes is different. It takes 2 minutes to decolorize 90% of alkaline dyes, while it takes 5 minutes for direct dyes. In contrast, azo dyes are more easily oxidized. Ozone can be used for the decolorization reaction of pigments by conjugating with ozone л- Explain the oxidation and decomposition of the electronic system. The common basic component in dyes is ortho hydroxy azo pigment. When these compounds react with ozone, the first step is for ozone to carry out electrophilic attacks on hydrazine ligands (which almost always exist in this form in solution). For example, the phenolphthalein of hydroxyphenylmethane pigments can be used as an indicator by reversibly opening and closing the lactone ring to produce color and color loss. Alkaline phenolphthalein easily reacts with ozone. Ozone undergoes a 1.3-addition reaction at the electron rich C=C bond, which can cut off the pigment skeleton and thus decolorize. When ozone reacts with typical triphenylmethylamine pigments such as malachite green, it simultaneously attacks the nitrogen and carbon frameworks (C=C bonds) at the dimethylamine site, which is similar to the reaction with phenolphthalein. When imine based pigments with C=N bonds react with ozone, ozone undergoes electrophilic reactions with C=N bond nitrogen atoms. Ozone simultaneously attacks the C=N bond and dimethylamino group to form an imidazoline ring, causing conjugation to be cut off and decolorization.
Second, with the attention to the tap water source environment and the reuse of secondary treated water in sewers, the decoloration of secondary treated water has been paid attention to. As for the color and taste caused by humus, the average chromaticity of water quality is 10 degrees. The maximum is 20 degrees. Such chromaticity can not meet the water quality standard of full removal by general coagulation and sedimentation and sand filtration processes, and may even exceed the worst standard. After ozone treatment, the chromaticity can be reduced to below L degree. Generally, the reason for coloring tap water is too much iron and manganese. If these metals are in a free state, they can be fully removed by conventional methods. If the raw water contains humus, sometimes chromium salt is formed, so it is quite difficult to treat it by conventional methods. Therefore, decoloration is also an important factor in introducing ozone treatment.

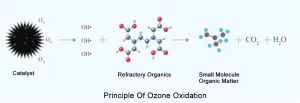
Decolorization mechanism of ozone: With the vigorous development of molecular biology, microecology extends ecology to molecular level. In fact, both protein and nucleic acid molecules are organic substances, and they are all composed of carbon, hydrogen, oxygen, nitrogen, phosphorus or sulfur (C, N, O, N, P or S). At the same time, the virus capsid is composed of many protein subunits, namely capsid particles. Each shell particle is connected by non-covalent bonds and symmetrically wound together, while protein is composed of multiple chains, and nucleic acid is composed of connected nucleotide chains. Among them,. OH, as a whole, is electrically neutral (R-OH), but if viewed from the inside of the group, part of it has more negative charges (such as oxygen atoms), because this part of the group (R-OH) has “extra” bonding electrons, so it is negatively charged; the other part has more positive charges (such as hydrogen atoms), and this part of the group lacks bonding. If another similar group approaches, the positive and negative charges attract each other to form a weak bond, which is called hydrogen bond. For example, hydrogen bonds are easy to form between groups of polypeptides or between base groups of nucleotides, and between base groups in DNA or RNA molecules. Although a single hydrogen bond is very weak, many hydrogen bonds are together, thus forming a tough cell wall of plant cells. Now look at ozone, which is a strong oxidant with high oxidation potential (2.07ev). All elements with high electronegativity can strongly attract electrons, oxidize each other and reduce themselves. As a result of oxidation, nucleic acid decomposition, protein disintegration, antigen denaturation, detection turned negative, and chromaticity faded out.

Ozone has a strong ability of decolorization, deodorization and odor removal, and it can produce odor such as chloric acid without adding chlorine agent. It has been reported that when ozone load is l-3mg/mgC (TOC, total organic carbon), almost all colors in water are removed. Generally, raw water has low color, smell and taste, so the dosage of ozone is only L-3 mg/L and the contact time is 10-15 minutes. According to the report, studies in China show that when the chroma of raw water is as high as 1,800-2,500 times and the COD is 1,100-1,800 mg/t, under certain conditions, the decolorization rate reaches 99% within 15 minutes, and the COD removal rate is close to 90%. The important factor affecting ozone decolorization is PH value. According to the research, when the PH value of wastewater decreases, the ozone consumption also decreases, so ozone decoloration is carried out at low pH.. In practical application, if ozone is used to treat groundwater, when iron and manganese are completely oxidized, the consumption ratio to ozone is 0.48mg ozone//mg Fe and 0.88mg ozone//mg Mn respectively. In high concentration Mn+2 water (1.10 mg/L), when the molar ratio of the two is 1:l, the oxidation rate is 95%, while in low concentration Mn+2 water ( A ratio of 0.5 removes more than 90% of the manganese.)