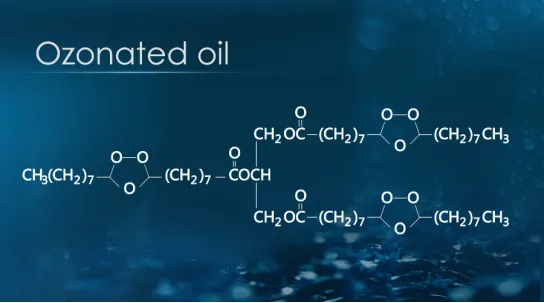
Ozone has strong decolorization, deodorization and odor removal capabilities, and it can avoid chlorine and other odors. It is reported that when the ozone load is l-3mg/mgC (i.e. TOC, total organic carbon), the color of water is almost completely removed; Generally, the raw water, color, smell and taste are low, so the dosage of ozone is only l-3mg/L, and the contact time is 10-15 minutes. It is reported that in China, when the color of raw water is up to 1800-2500 times and the COD is 1100-1800mg/t, under specific conditions, the decolorization rate can reach 99% within 15 minutes, and the COD removal rate is close to 90%. Then let’s take a look at the areas where the ozone generator is used for decolorization:
Ozone can decolorize all dyeing wastewater. Ozone can break the hair color and auxiliary groups of these dyes, so as to achieve the decolorization effect. However, the effect of ozone on various organic dyes is different. It takes 2 minutes to decolorize 90% of basic dyes and 5 minutes to decolorize direct dyes. In contrast, azo dyes are more easily oxidized. Ozone can be used to decolorize pigments from conjugation of ozone л- The oxidation decomposition of the electronic system is explained. The basic component of dyes is ortho hydroxy azo pigment. When these compounds react with ozone, first of all, ozone attacks the hydrazine backbone (almost always in this form in solution) in an electrophilic manner. For another example, phenolphthalein of hydroxybenzylmethane pigment can be used as an indicator through the reversible open and closed loop of lactone ring to generate color and discoloration. Alkaline phenolphthalein easily reacts with ozone. Ozone performs a 1.3 addition reaction at the electron rich C=C key, which can cut off the pigment skeleton and decolorize it. When ozone reacts with malachite green, a typical triphenyl JIA amine pigment, it simultaneously attacks the nitrogen and carbon skeleton (C=C bond) at the position of the diJIA amine, which is the same as when phenolphthalein reacts. When the azomethine pigments with C=N bond react with ozone, ozone reacts with the nitrogen atom with C=N bond in an electrophilic manner. Ozone attacks C=N bond and diJIA amino group at the same time to form oxazolidine ring, and the conjugation is cut off and decolorized.
With the attention paid to the tap water source environment and the reuse of sewage secondary treatment water, attention has been paid to the decolorization of secondary treatment water. As for the color and taste caused by humus, the average chromaticity of water is 10 degrees. Up to 20 degrees. Such chromaticity can not reach the water quality standard for full removal by the general coagulation sedimentation and sand filtration processes, and it may even exceed the bad standard. After ozone treatment, the chromaticity can be reduced to less than 1 degree. Generally, tap water is colored because of excessive iron and manganese content. If these metals are free, they can be fully removed by conventional methods. If the raw water contains humus and sometimes forms chromium salt, it is very difficult to treat it with conventional methods. Therefore, decolorization is also an important factor in introducing ozone treatment.